II. Physiology
- Images
- Calcium is the most common mineral found in the human body
- Bones and Teeth
- Metabolic processes
- Enzyme modulator (when bound to calmodulin)
- Neuromuscular Function and Nerve Impulses
- Blood Clotting Pathway
- Cell Movement
- Total body Calcium distribution
- Skeleton: 98%
- Circulating: 2%
- Free or ionized Serum Calcium (active): 50%
- Albumin-bound Serum Calcium (inactive): 50%
-
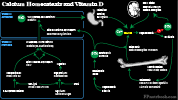
Parathyroid Hormone (PTH) modulates Serum Calcium (via Activated Vitamin D)
- PTH promotes renal activation of Vitamin D to 1,25-dihydroxycholecalciferol (activated Vitamin D)
- Calcium intestinal absorption is directly increased by 1,25-dihydroxycholecalciferol
- Intestinal Calcium absorption is the primary mechanism for regulating body Calcium
- Bone resorption increases with 1,25-dihydroxycholecalciferol
- Renal Distal convoluted tubule Calcium reabsorption increases with 1,25-dihydroxycholecalciferol
- Calcium intestinal absorption is directly increased by 1,25-dihydroxycholecalciferol
- PTH levels rise and fall inversely with Serum Calcium
- PTH rises in response to Low Serum Calcium (Hypocalcemia)
- PTH falls in response to High Serum Calcium (Hypercalcemia)
- PTH inhibits renal phosphate reabsorption
- Serum Phosphate would typically rise with Calcium reabsorption in gut, Kidneys and with bone resorption
- However, PTH's inhibition of renal phosphate reabsorption prevents Hyperphosphatemia
- PTH promotes renal activation of Vitamin D to 1,25-dihydroxycholecalciferol (activated Vitamin D)
-
Vitamin D effects vary by concentration
- Low concentrations of Vitamin D
- Promotes bone calcification
- Increases Calcium gastrointestinal absorption and renal reabsorption
- Persistent Vitamin D Deficiency may result in Rickets
- High concentrations of Vitamin D
- Functions in similar way to Parathyroid Hormone
- Increases bone resorption
- Low concentrations of Vitamin D
- Other factors affecting Calcium levels (less significant than PTH and Vitamin D)
- Calcitonin
- Calcitonin increases bone mineralization
- Calcitonin decreases Serum Calcium
- Blood pH
- Acidosis promotes Hypercalcemia
- Decreased Calcium plasma Protein binding (Calcium is displaced by excess Hydrogen Ion)
- Alkalosis promotes Hypocalcemia
- Increased Calcium plasma Protein binding
- Hyperventilation (and associated alkalosis) may result in transient Tetany from Hypocalcemia
- Acidosis promotes Hypercalcemia
- Calcitonin
III. Associated Conditions
- Hypercalcemia
- Hypocalcemia
- Rickets in children (Osteomalacia in Adults)
- Tetany
IV. References
- Goldberg (2014) Clinical Physiology, Medmaster, Miami p. 31