II. Physiology: Blood pH and Buffering Systems in General
- pH is a measure of Hydrogen Ion concentration
- pH=log (1/H)
- Where H is Hydrogen Ion concentration in gram moles per liter
- Neutral pH in water
- Hydrogen Ion is typically 10^-7 and is balanced by 10^-7 hydroxyl ion (OH)
- Body pH
- Normal arterial pH is 7.40 (Hydrogen Ion 40 nmole/L)
- Gastric Acid pH <3
- Pancreatic Secretion pH >8
- pH=log (1/H)
- Blood pH is normally maintained between 7.35 and 7.45 via buffers
- Weak acids buffer pH in a narrow range near 7.40
- Weak base (WB-) bound to Hydrogen Ions (H+) dissociate when a strong acid (SA) is present
- H+WB- => H+SA + WB-
- Extracellular buffers
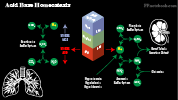
- Bicarbonate buffering system is the main extracellular buffer
- CO2 + H2O <=> H2CO3 <=> HCO3- + H+
- Intracellular buffers
- Intracellular Proteins, ammonia and phosphates
- Ammonia buffering system
- Ammonia (weak base) + Hydrogen Ion => Ammonium (NH3- + H+ => NH4)
- Glutamine is metabolized in renal tubule cells to Ammonium and bicarbonate
- Ammonium (NH4) is excreted in urine, while bicarbonate is reabsorbed in capillaries
- Phosphate buffering system
- Hydrogen phosphate + Hydrogen Ion => Dihydrogen Phosphate (HPO4 + H+ => H2PO4)
- Dihydrogen Phosphate (H2PO4) is excreted in urine
- Images: Acid Base Homeostasis
- Weak acids buffer pH in a narrow range near 7.40
- Low Electrolyte concentrations (Sodium, Potassium, chloride) decrease Hydrogen Ion concentration (Metabolic Alkalosis)
- Hyponatremia
- Increased Sodium reabsorption results in secretion of the hydrogen cation in exchange
- Bicarbonate absorption increases with Sodium reabsorption
- Aldosterone increases with Hyponatremia resulting in further Hydrogen Ion secretion
- Hypokalemia
- When Potassium is at normal level, it is excreted in exchange for Sodium
- When Potassium is reabsorbed in Hypokalemia, another cation is needed to exchange for Sodium
- In this case of Hypokalemia, Hydrogen Ion is secreted in exchange for Sodium absorption
- Hypochloremia
- Chloride is not available for reabsorption with Sodium from the renal tubule
- Another cation, in this case Hydrogen Ion, is secreted to balance negatively charged lumen
- Hyponatremia
III. Physiology: Bicarbonate buffering system (CO2-HCO3-)
- Bicarbonate buffering system equation
- CO2 + H2O <=> H2CO3 <=> HCO3- + H+
- Presence of strong acid shifts equation left toward CO2 + H2O
- Presence of strong base shifts equation right toward HCO3- and H+ ion
- Buffering Equation describes a balance between bicarbonate (HCO3-) and carbon dioxide (CO2)
- Water (H2O) combines with carbon dioxide (CO2) to form carbonic acid (H2CO3) catalyzed by carbonic anhydrase
- Carbonic acid (H2CO3) may freely dissociate with Hydrogen Ion (H+) to form bicarbonate (HCO3-)
- Under normal conditions blood bicarbonate (HCO3-) to dissolved CO2 ratio is 20:1
- pH and Hydrogen Ion (H+) are proportional to HCO3-/pCO2
- H+ : HCO3-/ pCO2
- Henderson-Hasselbach equation
- pH = 6.1 + log10 (HCO3-/(pCO2*0.03))
- where dissolved CO2 in plasma is only 3% of pCO2
- Hydrogen Ion increases (and pH decreases)
- Increased pCO2 (Respiratory Acidosis) OR
- Decreased HCO3- (Metabolic Acidosis)
- Hydrogen Ion decreases (and pH increases)
- Decreased pCO2 (Respiratory Alkalosis) OR
- Increased HCO3- (Metabolic Alkalosis)
- Homeostasis is maintained via respiratory (pCO2) and renal (HCO3-) mechanisms
- Lung function maintains pCO2 near 40 mmHg
- CO2 is a weak acid, and is the only acid excreted by the lung (all other acids are renally excreted)
- Brainstem responds to increased CO2 and H+ ion levels to increase Respiratory Rate reflexively
- Low oxygen level (O2) stimulates carotid and aortic body receptors (CN 9/10) to increase Respiratory Rate
- Renal Function maintains bicarbonate (HCO3-) near 25 mEq/L
- Bicarbonate is filtered by glomerulus and reabsorbed in renal tubules combined with Hydrogen Ion
- Total extracellular bicarbonate is 350 mEq for a 70 kg male
- Renal tubules excrete Hydrogen Ion
- Urine tends to be acidic (due to excess acid production over base production daily)
- Lung function maintains pCO2 near 40 mmHg
- Bicarbonate gains or losses impacts acidosis
IV. Physiology: Acid generation via metabolism
-
Carbohydrate and Fat Metabolism generates large amounts of CO2
- CO2 is quickly eliminated via respiration
-
Protein is metabolized into nonvolatile acid (fixed acid)
- Fixed Acid generated cannot be excreted as CO2
- Fixed Acid is buffered with bicarbonate to form carbonic acid
- Hydrogen Ion is renally excreted, maintaining bicarbonate for further buffering
V. Physiology: Renal Maintenance of Bicarbonate
- Bicarbonate is freely filtered by the glomerulus and reabsorbed by proximal tubule
- Glomerulus loses ~3600 meq bicarbonate daily (given 100 ml/min GFR) that must be reclaimed
- Nearly all bicarbonate is reabsorbed by the proximal tubule
- Bicarbonate levels above 26 mEq/L cannot be completely reabsorbed by proximal tubule
- Bicarbonate reabsorption (Metabolic Alkalosis) is increased with specific triggers
- Volume depletion (known as contraction alkalosis)
- Angiotensin II increased levels
- pCO2 increased levels (compensates for Respiratory Acidosis)
- Hypokalemia
- Renal Tubular Acidosis Type II results from defective proximal tubule reabsorption
- Causes Metabolic Acidosis through bicarbonate loss
- Hydrogen Ion renal excretion
- Primary mechanism for excreting fixed acid (see Protein Metabolism above)
- Proton Pump (ATP fueled)
- Pumps one Hydrogen Ion into collecting tubule
- Releases one bicarbonate to pass freely back into capillaries in the renal interstitium
- Renal Tubular Acidosis Type I (distal) results from defective Hydrogen Ion pump
- Glutamine Hydrolysis (proximal tubule)
- Renal key mechanism to compensate for acidosis (more than Hydrogen Ion excretion)
- Results in two outputs
- Ammonium (NH4+) which is excreted into urine
- Bicarbonate (HCO3-) which is absorbed by capillaries
VII. Pathophysiology
- Acidosis is due to one of 4 mechanisms
- Acid accumulation (e.g. Metabolic Acidosis with Anion Gap)
- Alkali loss (e.g. Diarrhea, Proximal RTA)
- Decreased renal acid accumulation (e.g. Renal Failure, Distal RTA)
- Carbon Dioxide accumulation (e.g. Respiratory Failure)
- Alkalosis is due to one of 3 mechanisms
- Excess CO2 exhalation in Respiratory Alkalosis (e.g. Hyperventilation)
- Alkali accumulation (e.g. iatrogenic with bicarbonate infusion)
- Acid loss (e.g. Vomiting)
VIII. Resources
IX. References
- Goldberg (2014) Clinical Physiology, Medmaster, Miami, p. 27-31
- Marino (2014) ICU Book, p. 587-99
- Preston (2011) Acid-Base Fluids and Electrolytes, p. 3-30
- Rose (1989) Clinical Physiology of Acid-Base and Electrolyte Disorders, p. 261-85