II. Mechanism: Energy Transfer
- Oxidative Phosphorylation (Electron Transport Chain)
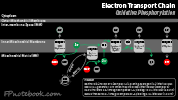
- Reaction cascade that converts Kreb Cycle energy from NADH and FADH to ATP
- Cytochrome based process within the mitochondria
- Oxygen dependent process (oxidation) coupled to phosphorylation of ADP to ATP
- When this process is uncoupled (e.g. uncoupler, hibernation), rare in humans, heat is generated
III. Types: Phosphoryl Transferring High Energy Molecules
- Adenosine Triphosphate (ATP/ADP)
- Adenine Nucelotide (sugar molecule with 3 attached phosphates, formed from Adenosine)
- Primary Function
- Primary energy currency of the cell (Energy Metabolism)
- RNA synthesis
- Other Functions
- ATP serves as a packet of energy currency (transfers energy via phosphorylation)
- Glycolysis (from Glucose, and from Fructose-6 Phosphate)
- ATP is generated from Adenosine Diphosphate (ADP) on phosphorylation by High Energy Molecules
- Glycolysis (from 1, 3 Biphosphoglycerate, and from Phophoenolpyruvate)
- Guanosine Triphosphate (GTP/GDP)
- Guanine Nucleotide (sugar molecule with 3 attached phosphates)
- Uridine Triphosphate (UTP/UDP)
- Uracil Nucelotide (sugar molecule with 3 attached phosphates, formed from Adenosine)
- Functions include Glycogen and Glucose Metabolism, as well as RNA synthesis
- Phosphocreatine (Creatine Phosphate)
- Phosphate source in skeletal Muscle, Myocardium as well as brain
- Phosphorylates ADP to ATP within the cell
- Phosphocreatine is metabolized to Creatinine and excreted in the urine, typically at a constant rate
- Phosphocreatine formation
- Guanidinoacetate (GAA) is sythesized in the Kidney from Arginine and Glycine (via AGAT enzyme)
- Non-phosphorylated Creatine forms by adding a methyl group to GAA in the liver (via GAMT enzyme)
- Phosphocreatine forms within Muscle Cells via Creatinine kinase activity
- https://en.wikipedia.org/wiki/Phosphocreatine
IV. Types: Hydrogen (electron) Transferring High Energy Molecules
- Nicotinamide Adenine Dinucleotide (NADH/NAD+)
- Transfers energy (electron, hydrogen) when it transitions from its reduced form (NADH) to oxidized form (NAD+)
- Key electron carrier for metabolic pathways including Krebs Cycle
- NADH maintains Hemoglobin's iron atoms in their reduced state (Fe++)
- Nicotinamide Adenine Dinucleotide Phosphate (NADPH/NADP)
- Transfers energy (electron, hydrogen) when it transitions from its reduced form (NADPH) to oxidized form (NADP+)
- NADPH Is produced in Hexose Monophosphate Shunt (HMP Shunt, Pentose Shunt), active throughout the body
- Functions
- NADPH reduces glutathione which protects RBCs against oxidative damage (e.g. superoxide radicals)
- Active in fat cells (Fatty Acid synthesis) and in liver cells (Cholesterol synthesis)
- Active in the Adrenal Cortex (steroid Hormone synthesis)
- Flavin Adenine Dinucleotide (FADH2/FADH)
- Transfers energy (electron, hydrogen) when it transitions from its reduced form (FADH2) to oxidized form (FAD+)
V. Types: Miscellaneous High Energy Molecules
- Biotin (Biotin-CO2/Biotin)
- Acetyl Coenzyme A (acetyl-CoA/CoA-Sh)
- Coenzyme A is an acetyl group donor in the Krebs Cycle, Fatty Acid biosynthesis and Amino Acid metabolism
- Activated Tetrahydrofolate (THF-C/DHF)
- Vitamin B9 (Folic Acid) derivative and a coenzyme in Amino Acid and Nucleic Acid synthesis
- Acts as single carbon donor in energy transfer (e.g. dUMP to TMP)
- Activated from Dihydrofolic acid (DHF) via dihydrofolate reductase (inhibited by Methotrexate)
- Thiamine Pyrophosphate (ThPP)
- Biologically active form of Thiamine (Vitamin B1)
- Cofactor in Carbohydrate Metabolism (oxidative decarboxylation)
- S-Adenosylmethionine
- Active sulfonium form of the Amino Acid, Methionine
- Acts as a methyl group donor in various reactions (enzymatic transmethylation)
- Formed from Methionine and ATP
- Uridine Diphosphate Glucose (UDP-Glucose/UDP)
- Carbohydrate Metabolism intermediate
- Glycogen precursor
- Polysaccharide generation (e.g. galactose, glucuronic acid)
- Lipopolysaccharide and glycosphingolipid synthesis
- Carbohydrate Metabolism intermediate
VI. References
- Goldberg (2001) Biochemistry, Medmaster, Miami, p. 4-12
